Current location: Home > PRODUCTS > Real Time PCR Series Products
PRODUCTS
(CE-IVD)
INTRODUCTION OF NTRK GENE
The NeuroTrophin Receptor Kinase (NTRK) family includes three proteins, TRKA, TRKB, and TRKC, encoded by the NTRK1, NTRK2, and NTRK3 genes, these proteins are normally expressed in neural tissues. Activation of TRK structures by NTRK gene fusions is considered as a pan-cancer oncogenic factor.
NTRK gene fusions are found in a variety of adult and pediatric solid tumors. In extremely rare tumor types such as infantile fibrosarcoma and secretory carcinoma, the frequency of NTRK fusion is more than 90%; in common cancers, the fusion frequency is between 0.1% and 2% [1].
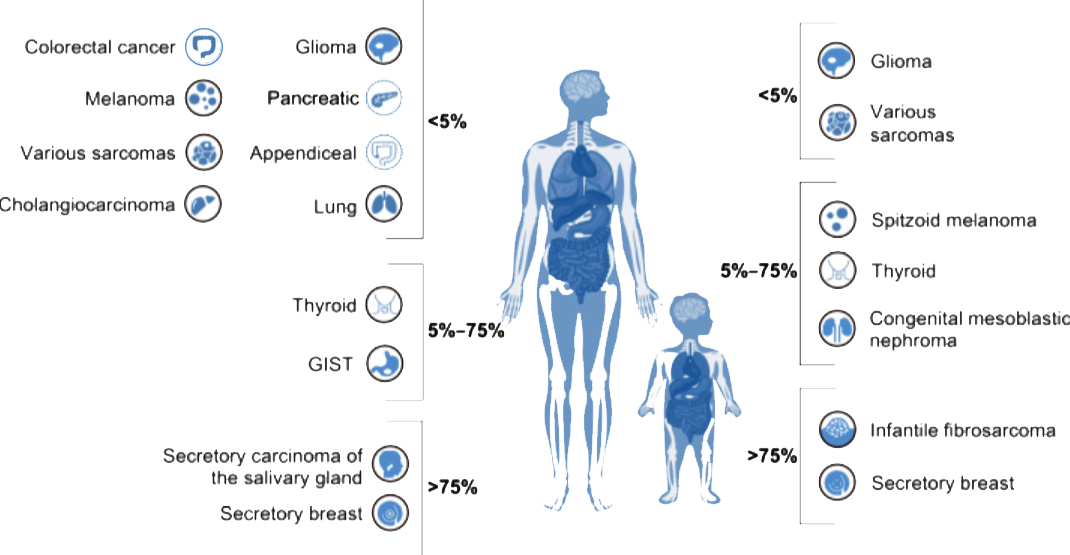
NTRK gene fusion profiles in adult and pediatric tumors[1]
NTRK AND TARGETED THERAPY
Although the frequency of NTRK fusions is low in common cancers, the efficacy of NTRK inhibitors is very obvious in patients with NTRK fusion-positive. The FDA approved larotrectinib and entrectinib in 2019 and 2020 for the treatment of patients with advanced solid tumors without alternative therapy or progression , who carry NTRK fusion-positive. This is the second and third pan-cancer FDA-approved tumor-targeted therapy. In clinical trials, the overall response rate (ORR) of larotrectinib and entrectinib reached 75% and 57%, this was achieved after 82% and 63% of the enrolled population had progressed on more than one systemic therapy [2].
Based on the above approval, the National Comprehensive Cancer Network has included NTRK inhibitors in several clinical diagnosis and treatment guidelines. The Chinese Society of Clinical Oncology (CSCO) guidelines for the diagnosis and treatment of non-small cell lung cancer (NSCLC) have also included NTRK inhibitors in the first-line treatment of stage IV NTRK fusion-positive NSCLC [3]. The detection of NTRK fusions is also recommended in guidelines such as CSCO colorectal cancer [4].
DETECTED GENES
Reaction Tube | Splicing Genes and Exons | NTRK Exon | Reaction Tube | Splicing Genes and Exon | NTRK Exon |
NTRK-1 | LMNA E2 | NTRK1 E10 | NTRK-3 | MPRIP E14 | NTRK1 E11 |
LMNA E10 | NTRK1 E10 | MPRIP E18 | NTRK1 E11 | ||
| LMNA E11 | NTRK1 E10 | MPRIP E21 | NTRK1 E11 | ||
SQSTM1 E5 | NTRK1 E9 | LMNA E6 | NTRK1 E1 | ||
TPM3 E8 | NTRK1 E9 | TPM3 E8 | NTRK1 E11 | ||
NACC2 E4 | NTRK2 E12 | SSBP1 E12 | NTRK1 E11 | ||
SQSTM1 E5 | NTRK2 E13 | STRN E3 | NTRK2 E16 | ||
VCL E16 | NTRK2 E13 | QKI E6 | NTRK2 E16 | ||
NTRK-2 | TFG E5 | NTRK1 E9 | NTRK-4 | EVT6 E4 | NTRK3 E14 |
TFG E6 | NTRK1 E9 | EVT6 E5 | NTRK3 E15 | ||
NFASC E21 | NTRK1 E9 | EVT6 E4 | NTRK3 E15 | ||
CD74 E8 | NTRK1 E9 | EVT6 E5 | NTRK3 E14 | ||
TPR E21 | NTRK1 E9 | EML4 E2 | NTRK3 E15 | ||
TPR E10 | NTRK1 E9 | TFG E6 | NTRK3 E1 | ||
IRF2BP2 E1 | NTRK1 E9 | ||||
BCAN E12 | NTRK1 E10 | ||||
BCAN E13 | NTRK1 E9 | ||||
LMNA E5 | NTRK1 E9 |
PRODUCT INFORMATION
Product Name | Technology | Pack Size | Instruments Validated | Sample Type |
Human NTRK Genes Fusion Detection Kit | Real-Time PCR assay | 20 Tests/Kit | ABI7500, ABI7300, ABI StepOne Plus, LightCycler480, Bio-Rad CFX96, etc. | Tumor tissue, Pleural and ascites |
DETECTION SIGNIFICANCE
Before systemic treatment in patients with advanced or metastatic solid tumors, NTRK gene fusion detection is performed for molecular typing to help guide treatments.
FEATURES & ADVANTAGES
1.Accuracy and Reliability:Use pre-load PCR tube to effectively avoid cross-contamination.
2.High Sensitivity:Sensitivity can reach as low as 100 copies in RNA.
3.Comprehensive Coverage:Covers the 32 most common variants of the NTRK.
4.Great versatility:Validated on the most common qPCR machines with stable results.
DETECTION PROCESS
1.Nucleic Acid Extraction
2.Set up qPCR
3.Amplification
4.Data Analysis
[1] J Mol Diagn. 2019 Jul;21(4):553-571.
[2] FDA official website data
[3] CSCO NSCLC Diagnosis and Treatment Guidelines 2023
[4] CSCO Colorectal Cancer Diagnosis and Treatment Guidelines 2023