Current location: Home > PRODUCTS > Real Time PCR Series Products
PRODUCTS
Human B-raf Gene Mutation Detection Kit
Real Time PCR
CE-IVD
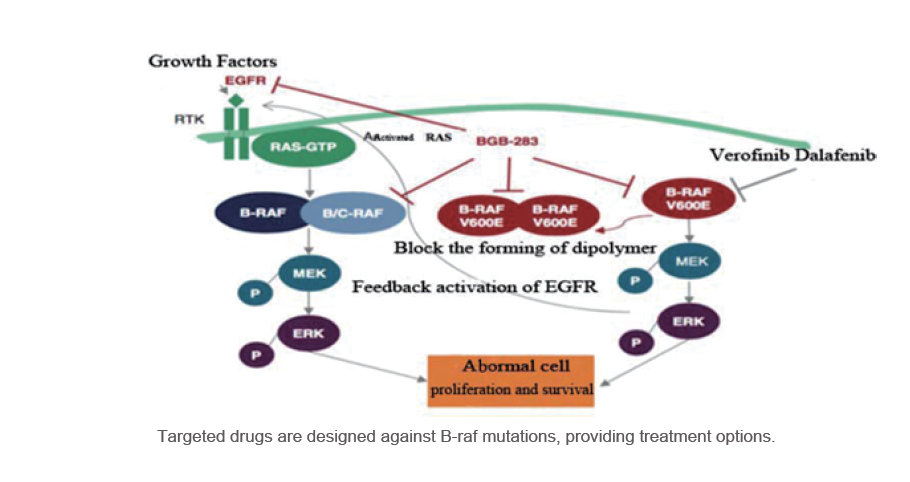
BRAF gene plays an important role in signal transduction involving cell proliferation, by regulating various kinds of cell physiological activities, such as growth, division , death etc.
BRAF gene is the second transmitter in the EGFR signaling pathway. When BRAF gene mutations occurs, the signal pathway will be permanently auto-activated, resulting in excessive cell proliferation and differentiation, thus, which may lead to EGFR-TKI drug ineffectiveness. Mutations of BRAF occurred in many cancers such as malignant melanoma, colorectal cancer, lung cancer, thyroid cancer, liver cancer,etc.
Clinical Practice Guideline for colorectal cancer (NCCN) (2019 Edition) suggested that for patients with metastatic colorectal cancer should be genotyped for B-raf and K-ras mutations.
Detection Item
| Gene | Cancer Type | Gene Status | Target Drug | Drug Indications |
| B-raf V600 | NSCLC | Mutation Type | Trametinib + Dabrafenib/ Vemurafenib | resistant |
| CRC(colorectal cancer) | mutation type | Encorafenib+ Cetuximab/ Panitumumab | sensitive | |
| melanoma | Wild Type | EGFR-TKI | Recommended to detect EGFR gene mutation | |
| wild type | Cetuximab/ Panitumumab | sensitive | ||
| mutation type | Vemurafenib+ Cobimetinib/ Binimetinib/ Trametinib + Dabrafenib | sensitive |
Performance Parameter
| Product Name | Technology | Product Specification | Matched Instrument | Sample Type |
Human B-raf Gene Mutations DetectionKit | PAP-ARMS® | 20tests/kit | StratageneMx3000pTM, ABI7500 etc. | tumor tissue Slice,ctDNA, pleural effusion |
Applicable People
● Patients with suspected Lynch syndrome can be checked by BRAF genetic detection.
●Can be used for the identification of diagnosis for thyroid cancer, assessing the prognosis for malignant melanoma and colorectal cancer.
● For patients treated with BRAF and EGFR inhibitors, detection of BRAF gene mutation status can be used to adjust diagnosis and treatments.
Technical Highlights
Accuracy and Reliability: Detection with closed tubes. Require no special treatment of the products.
Easy operation: One-step operation. 90 minutes of the whole process.
Good Reproducibility: Can be performed in the general PCR Labs. Results with good reproducibility can be easily obtained without special training.
Advanced Technology: R&D based on independent patented PAP-ARMS technology.
Detection Process
1.Nucleic Acid Extraction
2.Sample Adding
3.PCR
4.Report